Minimum Training Requirements
(1) CITI Certification
There is a need for minimum standards in training requirements to ensure the proper and ethical conduct of research on human subjects. This is to enable investigators and study team members to apply these ethical principles in the design, conduct and reporting of your research.
For NTU, we have partnered with the Collaborative Institutional Training Initiative (CITI) to offer the web-based training programme to cover various foundational topics on ethical research and human protection. Please click here to register for your CITI account using your NTU (or affiliated) email address, and choose one track (refer to image below) that is appropriate to your research.
Notes:
- CITI certification is required for all domains in NTU IRB, before the review of your ethics application can proceed.
- For undergraduate students, please also refer to our guidelines on “Students conducting research involving human subjects” for more details.

Guide to Updating CITI Profile with NTU/NIE Email Address
Login to CITI at https://www.citiprogram.org/index.cfm?pageID=14

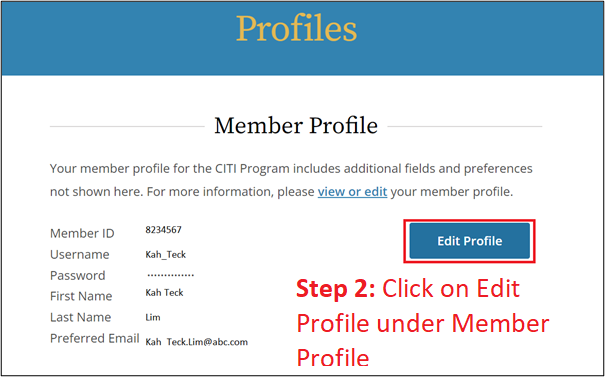
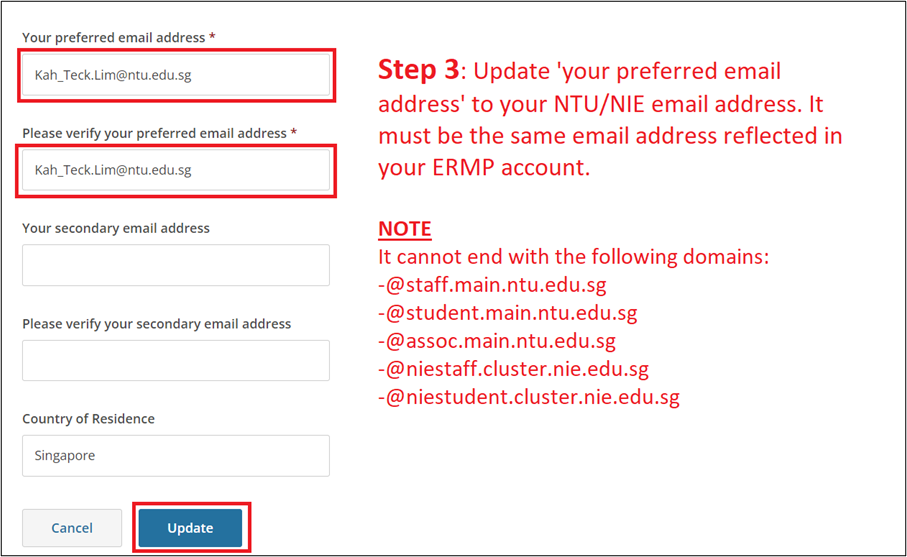

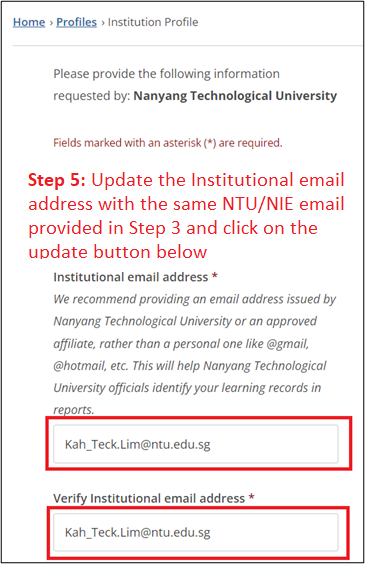
Important Notes
- Updates to your email address in CITI will not be immediately reflected on ERMP. It may take between 1 to 2 days for your training status to be updated on ERMP.
- Your NTU/NIE email address must appear in two locations of your CITI profile (see Steps 3 & 5).
- If you have more than one CITI account affiliated with NTU, ensure to update your NTU/NIE email only in the account which has your latest completion records. If more than one account has the same NTU/NIE email address, ERMP cannot identify which CITI account it has to sync with for your completion records.
- If ERMP still does still not reflect your CITI completion after the update, kindly review the above points before contacting [email protected] for assistance.
(2) HBRA Certification
Researchers conducting human biomedical research and tissue banking activities are required to obtain HBRA certification to ensure you are properly informed on HBRA requirements and penalties for any contraventions.
RIEO has developed an e-learning course, 'Human Biomedical Research Act (HBRA) Programme'.
- Staff learners may locate this course in Workday Learning by searching 'HBRA' under 'Browse Learning Content'.
- Student learners may locate this course in NTUlearn by searching for "HBRA' too.
Completion of this course by PI and team members is mandatory for projects under the scope of HBRA.

