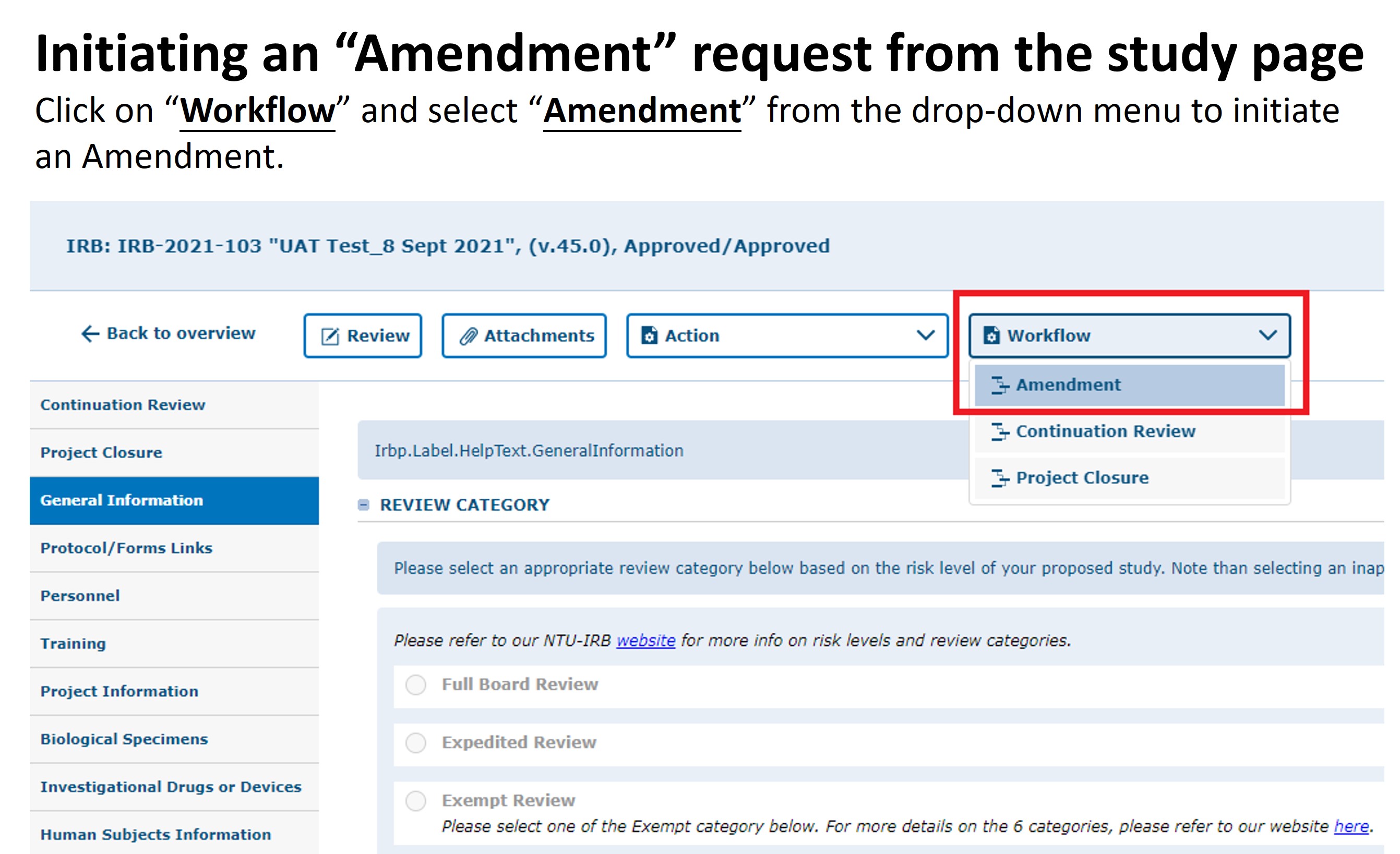
Amendments
Amendments to approved applications
PIs and study teams are required to adhere to their IRB-approved applications, including the protocol, target recruitment numbers, consent form, recruitment materials, data storage procedures, etc.
Any deviations from an approved protocol are considered Non-Compliances, and will be followed-up by NTU-IRB. PIs should also note that deviations from an approved protocol are considered as contraventions under the Human Biomedical Research Act (HBRA) and may be reportable to the relevant authorities .
If a modification to your approved application is required, PIs should submit an amendment to the IRB for review and approval before the change. (Amendments without prior approval are only allowed if there is an immediate safety risk to subjects.)
Exemptions to the Amendment Requirement
The following changes can be exempted from IRB approval and do not require an Amendment submission.
- Correction of typographical and/or grammatical errors that do not change the overall meaning of the text.
- Changes to colour palette of recruitment materials – e.g. flyer’s background colour.
It is the PI’s responsibility to ensure that all modifications comply with the IRB’s requirements. If you are unsure whether a modification requires review and approval, please consult with the IRB at [email protected].